HEU-EFS Interim Consortium Meeting
Advancing Premarket Programmes and Communication

[10 April 2024, online] – The HEU-EFS Consortium convened an online interim review meeting, marking a significant milestone in our journey towards harmonising Early Feasibility Studies (EFS) across Europe.
Key Outcomes:
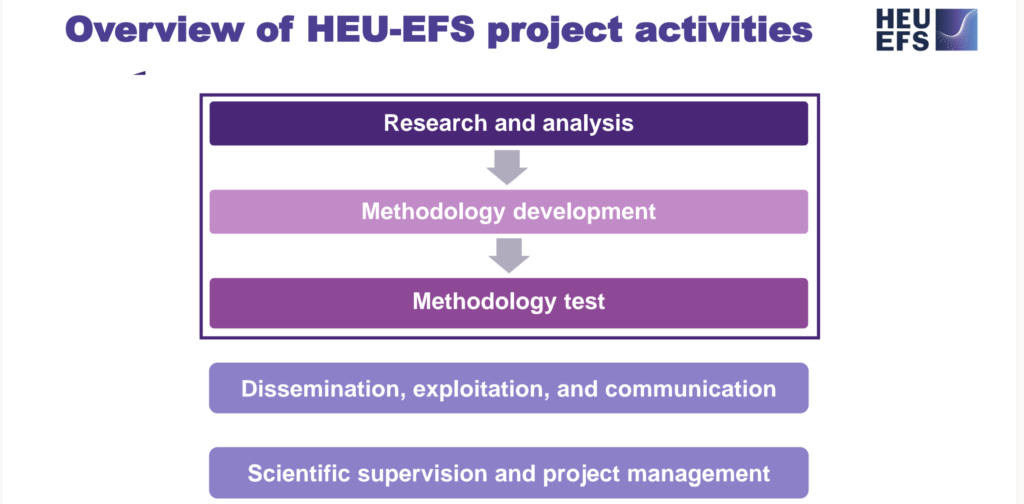
- Conducted detailed research of the regulatory framework and premarket programmes, pinpointing challenges to the implementation of Early Feasibility Studies (EFS).
- Strategies are being developed to address these challenges, aiming to streamline the process for healthcare innovations.
- The website is evolving into a key resource for disseminating research findings and engaging the healthcare community.
- Communication enhancements are underway to build a network of stakeholders vital for the EFS implementation across the EU.
- Tools for monitoring the performance of the EFS programme are in the development phase.
The collaboration and expertise of our consortium members were on full display as we worked together to address the challenges and opportunities within the EFS ecosystem. We are excited to share that our collective efforts are paving the way for a more efficient and effective European Health System.

A Video Guide to the HEU-EFS Project
In this video, you will hear from Rosanna Tarricone (SDA Bocconi) and Andrea Rappagliosi (Edwards Lifesciences Europe) as they explain the challenges and opportunities of medical device innovation in the European Union. You will also learn more about the HEU-EFS project’s objectives, activities, and expected impact.
At HEU-EFS, we believe that collectively, as a consortium of 22 institutions, SMEs and private companies, we can impact the health and well-being of people in Europe and beyond.https://youtube.com/shorts/f9EU57iv7eY?feature=share
Call for interest: Applications welcome for HEU-EFS’s Patient Advisory Group
Consortium partners are pleased to announce a call for representatives for a Patient Advisory Group (PAG) for the project ‘Harmonised Approach to Early Feasibility Studies for Medical Devices in the European Union (HEU-EFS)’. Applications remain open until 20 February 2024.
The Patient Advisory Group will discuss and provide recommendations for structured patient contribution to Early Feasibility Studies, in short EFS, to make more patient-centred medical devices. Early Feasibility Studies (EFS) are small-scale research studies or tests done in the very early stages of developing a medical device or treatment to see if it is practical, safe, and worth pursuing further. EFS help assess whether an idea or concept has potential before investing more time and resources into full-scale development and testing. The patient perspective in Early Feasibility Studies is highly important.
The PAG is meant to form a hub for patient centricity and engagement across HEU-EFS’s implementation. The aim is to recruit a maximum of ten representatives. Read more here

CSI Focus for Heart Failure
On December 4th, the HEU-EFS IHI initiative was presented at the annual leading conference in the field of device-based heart failure therapy, CSI Focus for Heart Failure (D-HF).
The HEU-EFS Consortium was invited to introduce the HEU-EFS project at a session dedicated to moving the field of heart failure focused devices forward. Moderated by Drs. Horst Sievert and William T. Abraham, the topic complemented the other speakers and was presented to a audience of clinicians and industry.
The program was positively received and enabled the expansion of the external advisory board to include representation of ethics committees.
More information on the conference and the session can be found here: https://www.csi-congress.org/conferences-courses/focus-workshops/csi-focus-d-hf/archive

Presentations
Implementing a Program for EarlyFeasibility Studies in Europe:Goldmine or Fool’s Gold? (ISPOR Europe, November 2025)
HEU-EFS Harmonised approach to Early Feasibility Studies for Medical Devices in the European Union (Clinical Investigations WG MD, 26 November 2025)
HEU-EFS Harmonised approach to Early Feasibility Studies for Medical Devices in the European Union (MDCG CIEPSE Working Group, 13 November 2025)
How to win and manage a Horizon project– the case of HEU-EFS (RIS4SEB Consortium meeting, Milan, 9 September 2025)
HEU-EFS Harmonised approach to Early Feasibility Studies for Medical Devices in the European Union - EU Health Technology Assessment: Advent of a new era of collaboration (HTA Conference, 2 July 2025)
Fostering an Enabling Landscape (FutureTrials 25, 17 June 2025)
Marta Bragagnolo and Yasemin Zeisl Presentation on The patient perspective: Involvement of patients in translational research and clinical investigations (EORS, 16 – 19 June 2025)
PD Dr. med. Thomas M. Randau Presentation on Improvement through Innovation – How much evidence do you need to adopt? (EORS, 16 – 19 June 2025)
Susan Partridge Presentation on Clinical evidence generation for high-risk orthopaedic medical devices under MDR (EORS, 16 – 19 June 2025)
Ana Matos Machado Presentation on Clinical evidence generation for high-risk medical devices under MDR (EORS, 16 – 19 June 2025)
Sebastian Kuhn Presentation on Early Clinical Investigations for Digital Health Technologies (EORS, 16 – 19 June 2025)
Marta Kerstan (DePuy Synthes) and Alexandra Poulsson (NIPH) Presentation on European Orthopaedic Research Society Annual meeting (EORS, 16 – 19 June 2025)
Challenges of pre-market clinical investigations of medical devices: a multi-stakeholder perspective – Issue Panel Bridging the Gap in Early-Stage Clinical Research: Lessons for EU Stakeholders from the US FDA Early Feasibility Studies Program (ISPOR 2025, Montréal, 15 May 2025)
EFS Advantages in Device Development (ISPOR 2025, May 2025)
Empowering European Innovators With Early Feasibility Studies (MedTechForum 2025, 14 May, 2025)
HEU-EFS Harmonised approach to Early Feasibility Studies for Medical Devices in the European Union (MDCG Clinical Investigation and Evaluation Working Group, 8 April 2025)
The role of patients in the early feasibilitystudies of medical devices, PEDs and PROs – Engaging patients in the development of an Harmonised Approach to Early Feasibility Studies for Medical Devices in the European Union (ESC, 20 February, 2025)
US vs. EU: 10 Years of Experience with EFS vs. a Program Starting to Emerge in Europe (Privileged Talk, Milan, 18 March 2025)
Marta Bragagnolo (GHH) on Patient Involvement Webinar in Early Feasibility Studies (EUPATI Webinar, 28 January 2025)
HEU-EFS Harmonised approach to Early Feasibility Studies for Medical Devices in the European Union – Progress and preliminary results (MDCG Clinical Investigation and Evaluation Working Group, 15 November 2024)
HEU-EFS Harmonised approach to Early Feasibility Studies for Medical Devices in the European Union (Medtech Europe, 24 September 2024)
HEU-EFS Harmonised approach to Early Feasibility Studies for Medical Devices in the European Union – Engagement of National Competent Authorities (CIE, 22 August, 2024)
HEU-EFS Harmonised approach to Early Feasibility Studies for Medical Devices in the European Union – Overview (MedTech Europe, 16 July 2024)
Harmonized Approach for Medical Devices in the EU to EFS (HTAi 2024 Annual Meeting, 17-19 June 2024)
Health Technology Assessment: Methods, Tools, and Specific Criteria Applicable to Innovative Medical Devices (GERONTE Project Leuven March 2024)
HEU-EFS Harmonised approach to Early Feasibility Studies for Medical Devices in the European Union (MedTech Europe Cardiovascular Sector Group, 8 March 2024)
HEU-EFS Harmonised approach to Early Feasibility Studies for Medical Devices in the European Union (MDCG Clinical Investigation and Evaluation Working Group, 4 March 2024)
HEU-EFS – Harmonised approach to Early Feasibility Studies for Medical Devices in the European Union (Info day Horizon Europe European Partnerships, 26 February 2024, Milano)
Setting up a harmonized methodology to promote uptake of early feasibility studies for clinical and innovation excellence in the European union: What value(s)? Presentation at ISPOR Europe (14 November 2023, Copenaghen)
Project Slide Deck (14 April 2024)
Harmonized Approach to EFS for Medical Devices in the EU (CSI Devices for Heart Failure (DHF), 9 December 2023)
Harmonized Approach to Early Feasibility Studies for Medical Devices in the European Union (HEU-EFS) presented by Prof. Giuditta Callea, PhD at Wired Health (13 March 2024)
Posters
National Competent Authorities’ Experiences With Early Feasibility Study Assessment: Promoting Dialogue, Supporting Sponsors, and Fostering Shared Learning to Harmonize the EU Framework presented at Ispor Europe 2025 (11 November 2025)
Are Early Feasibility Studies an orphan approach in the regulatory pipeline for medical devices clinical maturity? by Adrián Valledor presented at RAPS Euro Convergence 2025 (16 May 2025)
Challenges of pre-market clinical investigations of medical devices: a multi-stakeholder perspective presented at Ispor US 2025 (13 May 2025)
Use of Early Feasibility Studies to Inform Development of Medical Devices presented at ISPOR Europe (20 November 2024)
Global Assessment of Pre-Market Approval Pathways for Medical Devices: Highlighting the Need for Harmonization across 55 Jurisdictions by Francesco Malandrini presented at ISPOR Europe 2024 (17 November 2024)
Press Releases
[30 January 2024, Milan]
Improving patient access to innovative medical technologies in the European Union
HEU-EFS Project Video Guides
Executive Summary
WP1 – Early Feasibility Studies and pre-market clinical investigation pathways: context, facilitators and barriers
Leader: Giuditta Callea (SDA Bocconi)
Co-leader: Marta Kerstan (DePuy Synthes)
WP2 – The EU regulatory framework and EFS
Leader: Tom Melvin (Trinity College Dublin)
Co-leader: Nicolas Martelli (Assistance Publique – Hôspitaux de Paris)
WP3 – Methodology development: rationale, processes and procedures
Leader: Alexandra Poulsson (Norwegian Institute of Public Health)
Co-leader: Nataliya Deych (Edwards Lifesciences Europe)
Co-leader: Carlo Federici (SDA Bocconi)
WP4 – Methodology development: evidence requirements, data, and statistical tools
Leader: Monica Tocchi (Meditrial Srl)
Co-leader: Alessandra Denaro (Abbott)
Co-leader: Keshav Kohli (Abbott)
WP 5 – Methodology development: EU EFS Program monitoring system
Leader: Giuditta Callea (SDA Bocconi)
Leader: Rosanna Tarricone (SDA Bocconi)
Leader: Francesco Malandrini (SDA Bocconi)
Co-leader: Antonio Balsamo (Agenas)
Co-leader: Gabriella Di Santo (Agenas)
Co-leader: Alessandra Lo Scalzo (Agenas)
Co-leader: Marco Marchetti (Agenas)
WP 6 – Methodology development: ethical and legal aspects
Leader: Giuditta Callea (SDA Bocconi)
Leader: Rosanna Tarricone (SDA Bocconi)
Co-leader: Irene González Blanquer (Edwards Lifesciences Europe)
Co-leader: Pierre-Alain Dumas (Edwards Lifesciences Europe)
WP7 – Testing the HEU-EFS methodology: pilot use-case
Leader: Andrea Rappagliosi (Edwards Lifesciences Europe)
Leader: Alessandra Denaro (Abbott)
Leader: Karine Miquel (Abbott)
Co-Leader: Barbara Indino (Medtronic)
Co-Leader: Manon Gielkens (Medtronic)
Co-Leader: Carlo Trani (Fondazione Policlinico Universitario Agostino Gemelli IRCCS)
Patient Advisory Group members
Phil Collis – Cardiovascular disease patient
Lucía Feito Allonca – Type 1 diabetes patient
Massimo Russo – Cardiovascular disease patient
Russell Wheeler – Carer/advocate for an eye condition patient
Dieter Wiek – Axial Spondyloarthritis patient
Yasemin Zeisl – European Patients‘ Forum
Marta Bragagnolo – Global Heart Hub
HEU-ESF Pilot Training Videos
This section includes ten training videos developed for stakeholders participating in the HEU‑EFS pilot process. The videos outline the key elements of the HEU‑EFS framework that stakeholders will follow throughout the pilot, including key definitions for EFS, eligibility criteria, requirements for clinical sites and investigators, stakeholder roles, overviews of templates and checklists for the clinical investigation plan, informed consent form, master clinical trial agreement and insurance agreement, strategies for patient involvement, and a complete description of the pilot process.
What is an EFS and when should an EFS be performed
Definitions of EFS and low- vs high-risk medical device
Presenter: Maria Luisa Buzelli (UB)
When is an EFS relevant – Eligibility Criteria
Description of the eligibility criteria for medical device technologies, patient population, and level of preclinical evidence to participate in the HEU‑EFS pilot
Presenter: Kevin Drisko (Edwards Lifesciences)
Clinical sites and clinical expertise for EFS pilots
Overview of requirements for clinical sites to participate in the HEU-EFS pilot, such as those related to regulatory competency, good clinical practices, equipment, and clinical staff experience
Presenter: Nicolas Martelli (APHA)
HEU-EFS proposed pilot framework
Overview of the elements of the HEU-EFS framework, including Contact Point, Early Dialogue, Submission, Validation, and Review
Presenter: Alexandra Herborg Cornelius Poulsson (NIPH)
An introduction to the actors involved in an Early Feasibility Study
Description of responsibilities of key actors (i.e., sponsor, CRO, regulatory authority, EC, investigational site, Principal Investigator) involved in the HEU-EFS pitot process
Presenter: Tom Melvin (Galway)
Clinical Investigation Plan Template – Methodology development, evidence requirements, data, and statistical tools
Description of development and content of Clinical Investigation Plan template and checklist
Presenter: Monica Tocchi (Meditrial)
Informed Consent Form, Master Clinical Trial Agreement and Insurance Agreement
Description of supporting documents (i.e., Informed Consent Form, Master Clinical Trial Agreement and Insurance Agreement) during the HEU‑EFS pilot process
Presenter: Francesco Malandrini (UB)
Key Performance Metrics
Description of the approach used to collect data required to calculate Key Performance Indicators (KPIs) of HEU-EFS pilots
Presenter: Federico Facciolo (UB)
When and how to involve Patients
Description of patient involvement strategies to be used before, during, and after the EFS
Presenter: Yasemin Zeisl (EPF)
HEU-EFS pilot process
Description of the key process steps of the HEU-EFS pilot process, including Expression of Interest, Information Call, Online Form, Prioritization, Training on HEU-EFS, and application to NCA
Presenter: Giuditta Callea (UB), Manon Gielkens (Medtronic), Fanny van der Loo (Edwards Lifesciences)
Publications
Articles
From fragmentation to framework: why early regulatory advice is critical for European medical device innovation.
Melvin, T., Callea, G., & Rappagliosi, A. (2026). Expert Review of Medical Devices, 1–3. https://doi.org/10.1080/17434440.2026.2640201
Regulatory readiness for innovation: a mixed-methods study of national competent authority professional and organizational capacities in the context of pre-market clinical investigations and early feasibility studies
Geraghty, M., Malandrini, F., Callea, G., McDonnell, A., Martelli, N., Tangila Kayembe, O., Kuhn, S., Herborg Cornelius Poulsson, A., & Melvin, T.
Expert Review of Medical Devices, January 2026, doi.org/10.1080/17434440.2025.2594460
Stakeholder Perspectives on Early Feasibility Studies for Digital Health Technologies in the European Union: A Qualitative Interview Study
Peseke M., Michaelis I., Kayembe O., Geraghty M., McDonnell A., Zurlo F., Oftring Z., Martelli N., Melvin T., Kuhnn S.
JMIR – Journal of Medical Internet Research, JMIR Publications Inc, October 2025, 10.2196/77982
Conference Proceedings
MT13 Challenges of Pre-Market Clinical Investigations of Medical Devices: A Multi-Stakeholder Perspective
Callea, Giuditta et al., Value in Health, 28 (6), S268, July 2025, https://doi.org/10.1016/j.jval.2025.04.1123
MT58 Use of Early Feasibility Studies to Inform Development of Medical Devices
C Federici, FL Zurlo, R Tarricone, G Callea
Value in Health, 27 (12), S497, December 2024, 10.1016/j.jval.2024.10.2528
HPR98 The EU Regulatory Framework for Medical Device Early Feasibility Studies: What Do We Know to Date?
T Melvin, N Martelli, S Kuhn, O Tangila, Kayembe, I Michaelis, M Peseke, A McDonnell, F Murphy∙ M Geraghty, H Banks, G Callea, R Tarricone
Value in Health, 27 (12), S294, December 2024, 10.1016/j.jval.2024.10.1524
HPR67 Global Assessment of Pre-Market Approval Pathways for Medical Devices: Highlighting the Need for Harmonization Across 55 Jurisdictions.
Malandrini FB, Callea G, Tarricone R, Kerstan M, Sampietro-Colom L, Valledor A, Balsamo A, Catania D, Vainieri S, Tocchi M, Tangila Kayembe O, Melvin T, Poulsson A, Kuhn S, Rappagliosi A, Auclair P, Gielkens M, Santin C, Roijers R, Kvistgaard Jensen L
Value in Health, 27 (12), S288, December 2024, 10.1016/j.jval.2024.10.1494
Press release: Improving patient access to innovative medical technologies in the European Union
The link to the print version of the press release
European consortium to develop a “Harmonized Approach to Early Feasibility Studies for Medical Devices in the European Union” (HEU-EFS)
[30 January 2024, Milan] – The HEU-EFS Consortium has announced its creation. This four-year project, funded by the Innovative Health Initiative under the Horizon Europe Framework Programme and involving 22 public and private organisations across Europe (including EEA and Switzerland), aims to improve the early evidence generation pathway for medical device innovators. This will contribute to foster access to innovative technologies for patients with unmet needs, strengthen clinical development excellence, increase attractiveness of the EU, and support healthcare system’ efficiencies.
Technological innovation in health care continues to expand to respond to increasing patient needs. However, the innovators’ journey to bring such innovations to patients in a timely and harmonised manner has become long and complex. This is partly due to rising R&D costs, ever-stricter European regulations as well as a lack of harmonised approaches in Member States for early evidence generation.
This creates uncertainty for innovators and a decline in attractiveness of Europe, with delays in bringing these breakthrough technologies to the EU market. Ultimately patients in Europe are missing out on the latest medical technology innovations that could improve their prognosis and quality of life.
HEU-EFS aims to contribute to revering this trend. It will focus on the development of a Harmonised Approach to EFS for Medical Devices in the European Union. EFS are small clinical studies designed to gain early insights into an innovative medical technology early in the development process, before starting a larger clinical trial.
EFS represent a crucial first step in the evidence-generation pathway, allowing innovators to test their potentially breakthrough innovations and build a robust business case, and the healthcare systems to better accompany the introduction of those new innovations that could bring value to patients in need.
Although the possibility to undertake EFS is formally recognized by the international standard, in the EU there is no procedure, standard or guideline dedicated to this type of study. This means that EFS are barely used in Europe, and early evidence generation is taking place elsewhere in the world.
HEU-EFS will develop and validate a robust, unified approach to allow early insights into technology evidence generation and to make the EU a conducive environment to undertake EFS. This will contribute to improving efficiency and effectiveness of product development, and ensure that the EU can attract investments into patient-centric, end-user efficient innovations that bring value to the healthcare system.
“The HEU-EFS program is a crucial step for positioning the EU as a worldwide epicenter for cutting-edge technologies. This strategic stance will draw investments in research and development, ideas, and expertise, thereby ensuring swift access to top-tier healthcare for patients”, says Rosanna Tarricone, Associate Professor at the Department of Social and Political Sciences and Associate Dean for Government, Health and Non-Profit Division SDA Bocconi, Bocconi University.
The consortium is funded by a €19 million grant from the Innovative Health Initiative (IHI), a public-private partnership between the European Commission and European life science industry associations. The project is coordinated by Bocconi University and the industry partners are led by Edwards Lifesciences.Engagement of wide range of stakeholders – universities, hospitals, contract research organizations, patient associations, health technology assessment agencies, MedTech companies and start-ups, regulatory authorities, notified bodies, professional associations and members of ethics committees – will drive a wide acceptability of the framework across countries and sectors.
According to Andrea Rappagliosi, Senior Vice President Public Affairs EMEA, Canada and LATAM at Edwards Lifesciences, “The new HEU-EFS EU program will allow innovative MedTech companies to significantly contribute to improve access for patients in Europe. This will contribute to a triple win: a healthier population, a more efficient health and hospital system, and a more attractive and competitive EU”.
HEU-EFS KICK-OFF MEETING
30-31st OCTOBER 2023, MILAN: the launch of an innovative EU initiative to foster innovation in the MedTech sector.
In an exciting and collaborative atmosphere, the kick-off meeting of the “Harmonized Approach to Early Feasibility Studies for Medical Devices in the European Union” (HEU-EFS) project took place on October 30-31 at SDA Bocconi campus in Milan, marking the initiation of a ground-breaking initiative aimed at fostering patient-centred innovation and cooperation within the EU MedTech sector.
TheHEU-EFS project will facilitate access to innovative medical devices (MDs) in the European Union (EU) by developing a shared and breakthrough program dedicated to Early Feasibility Studies (EFS), small clinical studies designed to gain early insights into an innovative medical technology during the development process. This program will contribute to making the EU an attractive jurisdiction for innovative MDs developers and accelerate patients’ access to the best possible care. This 4-year project secured €10.53 million in funding from the European Commission and €8.47 million from industry partners, for a total of €19 million, as part of the Innovative Health Initiative (IHI). The commitment of IHI – a public-private partnership between the European Commission and major industry associations – and the partners reflects the relevance of EFS and the common understanding of the need to evolve toward a collaborative, system-wide approach to support innovation in the EU.
The kick-off provided an opportunity to bring together for the first time the diverse group of stakeholders composing the consortium, for a total of 22 partners from 13 different countries, and representatives of the Advisory Board, and to establish a solid foundation for successful collaboration.
The kick-off was opened by Rosanna Tarricone (Associate Professor at the Department of Social and Political Sciences Bocconi University and Associate Dean for Government, Health and Non-Profit Division SDA Bocconi) as Project Coordinator, Andrea Rappagliosi (Senior Vice President Public Affairs, EMEA, Canada and Latin America, Edwards Lifesciences) coordinator of the private part of the consortium and Geraldine Joanny (IHI Scientific Project Officer), who emphasized the significance of this collaboration among public and private stakeholders in tackling EU health challenges.
After a roundtable presentation of all the partners, an overview of the project’s background, goals, timelines, and expected outcomes was provided, followed by a deep dive into the specificities of the 9 WPs composing the project.
The meeting facilitated fruitful discussions on the relevance of early clinical evidence generation as a first step in a lifecycle approach. It was also an occasion to clarify legal and administrative rules, discuss the distribution of responsibilities among the consortium members, and emphasize the importance of continuous and effective communication. A key highlight was the discussion on how to engage patients in the project to effectively implement patient centricity in decision-making.
The enthusiasm and collaborative spirit witnessed during the kick-off meeting set a positive tone for the journey ahead, promising breakthroughs and advancements that will elevate healthcare innovation within the EU.
